Atomic structure
'We think there is color, we think there is sweet, we think there is bitter, but in reality there are atoms and a void.'
|
Key ideas and concepts
- Atoms are building blocks of matter made of 3 types of subatomic particles: protons, neutrons, and electrons.
- Protons and neutrons are packed in the centre of atoms, called the nucleus, and concentrate most of the mass of an atom.
- Negatively charged electrons are attracted to the positively charged nucleus. They move rapidly in the area around it. Their contribution to the mass of an atom is negligible.
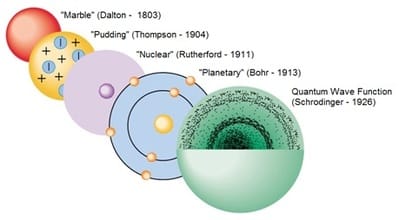
1. Let's start with a bit of history
- 1803: John Dalton introduced a new form of the ancient Greek idea of atoms. He modelled them as hard balls.
- 1897: J.J. Thomson discovered the electron and suggested the 'plum pudding' model of the atom.
- 1911: Ernest Rutherford proposed a new model in which the positive charges in the atom were concentrated in a small central core or nucleus, disproving Thomson's plum pudding model.
- 1914: Niels Bohr modified Rutherford's model by introducing the idea of energy levels, where the electrons move rapidly around the nucleus in fixed orbits, like planets orbiting the sun.
- 1926: Schroedinger developed the wave function that describes the position and state of an electron in an atom in terms of probability, showing that electrons can no longer be thought as orbiting the nucleus at a well defined distance.
- Since 1926, we think of an atom as a positively charged nucleus surrounded negatively charged electrons occupying well-defined energy levels. An energy level can be thought as a region of space where the electrons in that level are most likely to be found, hence the term 'electron cloud' to describe the distribution of electrons in atoms.
2. The different parts of an atom
Three types of sub-atomic particles can be found in an atom:
- Proton: positively charged particle, located in the nucleus.
- Neutron: no electrically charge, similar mass to the proton. Located in the nucleus.
- Electron: negatively charge particle. Arranged in shells (energy levels) around the nucleus.
3. atomic number
- The number of protons in the nucleus of an atom is called atomic number.
- Protons and neutrons are collectively called nucleons.
- The number of protons+neutrons is called mass number.
4. the size of atoms
- An atom is roughly a million times smaller than the thickest human hair. That is smaller than a billionth of a meter.
- The nucleus of an atom is even smaller than an atom: If an atom was enlarged to the size of a football stadium, the nucleus would be the size of a marble in the centre of the field and the electrons would be moving rapidly around the stadium, with nothing existing in-between.
- In other words, the diameter of a nucleus is about 10 000 to 100 000 smaller than the diameter of an atom, justifying the statement that an atom, and therefore matter, is mostly empty space (about 99.999999999 %).
- To understand this number, imagine that you could remove the empty space from the atoms of all people on Earth. The entire human race could then fit in the volume of a sugar cube!