States of matter
Key ideas and concepts
- A state of matter is one of the distinct forms that matter takes on. Three states of matter are observable in everyday life: solid, liquid, gas.
- Changing between states of matter involves adding or removing heat.
- The particle model is useful to explain the behaviour of and differences between solids, liquids and gases.
1. the different states of matter
Everything around you and making you is made up of matter. On Earth, matter can be found under 3 different physical forms called the states of matter. Since you were a baby, you learnt that "things" occur as solids, liquids, or, less obviously, as gases. The main differences between these states are described below.
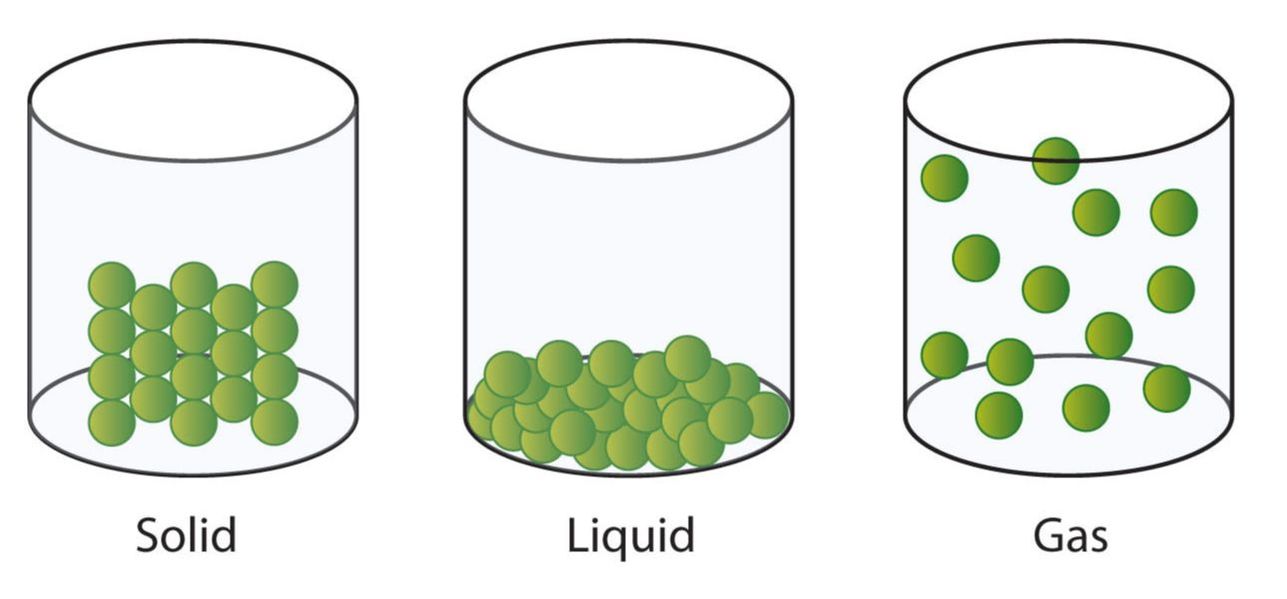
- Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place.
- Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely.
- Matter in the gaseous state has both variable volume and shape, adapting both to fit its container. Its particles are neither close together nor fixed in place.
DID YOU KNOW?
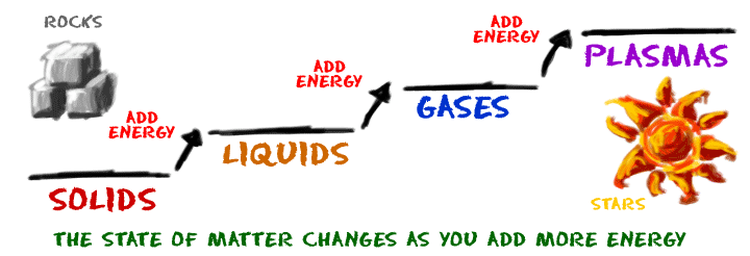
A fourth state is also commonly observed in everyday life, the plasma state. Matter in the plasma state has variable volume and shape, but as well as neutral atoms, it contains a significant number of ions and electrons, both of which can move around freely. Maybe surprisingly, plasma is the most common form of visible matter in the universe as matter making up all stars are in the plasma state. On Earth, the plasma state occurs in lightnings, neon and fluorescent tubes, cathode ray tubes and plasma spheres. The energy stored in a plasma state is released as light and heat.
A fourth state is also commonly observed in everyday life, the plasma state. Matter in the plasma state has variable volume and shape, but as well as neutral atoms, it contains a significant number of ions and electrons, both of which can move around freely. Maybe surprisingly, plasma is the most common form of visible matter in the universe as matter making up all stars are in the plasma state. On Earth, the plasma state occurs in lightnings, neon and fluorescent tubes, cathode ray tubes and plasma spheres. The energy stored in a plasma state is released as light and heat.
2. changes between states of matter
Most pure substances can exist in any of these three states and it is possible to change from one state to another by adding or removing heat.
The transition between the solid and liquid states and between the liquid and gas states of a substance require the input of heat energy. Think about it: you need to heat up water to bring it to a boil. However, there is another way to boil a liquid a room temperature by decreasing the pressure of the surrounding air.
The transition between the solid and liquid states and between the liquid and gas states of a substance require the input of heat energy. Think about it: you need to heat up water to bring it to a boil. However, there is another way to boil a liquid a room temperature by decreasing the pressure of the surrounding air.
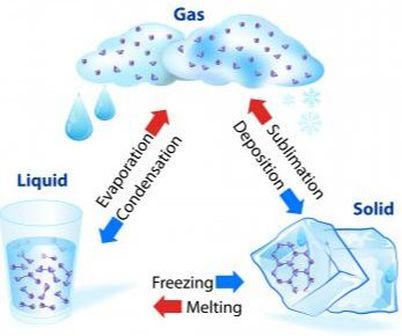
Any of these changes of state have their own name as shown below.
The process of vapourization is sometimes called evaporation or boiling, as in your textbook on page 108.
The process of freezing is also know as solidification.
The melting point of a solid is the temperature at which all of the solid melts. During melting, the temperature of the solid stays the same and equal to the melting point. For example, the melting point of the element gallium is 30 oC. Can you imagine what interesting property this low melting point gives to gallium? Other examples are the melting point of the element hydrogen, -259 oC, and that of the element uranium, 1135 oC. These values were obtained from an online Interactive Periodic Table.
In the same way, the boiling point of a liquid is the temperature at which the liquid boils. During boiling, the temperature of the liquid does not change and is equal to the boiling point. For instance, liquid nitrogen boils at -196 oC while gold boils at 2856 oC.
The condensation point of a gas is equal to the boiling point of the corresponding liquid. For example, since mercury boils at 357 oC, it can be predicted that its condensation point is 357 oC as well.
Some solids do not melt but sublime: they become a gas without forming a liquid first. Solid substances such as dry ice, diamond and graphite have no known liquid state. Heat up a diamond hot enough and it will literally evaporate!
Challenge: Answer in your notebook the following questions
The process of freezing is also know as solidification.
The melting point of a solid is the temperature at which all of the solid melts. During melting, the temperature of the solid stays the same and equal to the melting point. For example, the melting point of the element gallium is 30 oC. Can you imagine what interesting property this low melting point gives to gallium? Other examples are the melting point of the element hydrogen, -259 oC, and that of the element uranium, 1135 oC. These values were obtained from an online Interactive Periodic Table.
In the same way, the boiling point of a liquid is the temperature at which the liquid boils. During boiling, the temperature of the liquid does not change and is equal to the boiling point. For instance, liquid nitrogen boils at -196 oC while gold boils at 2856 oC.
The condensation point of a gas is equal to the boiling point of the corresponding liquid. For example, since mercury boils at 357 oC, it can be predicted that its condensation point is 357 oC as well.
Some solids do not melt but sublime: they become a gas without forming a liquid first. Solid substances such as dry ice, diamond and graphite have no known liquid state. Heat up a diamond hot enough and it will literally evaporate!
Challenge: Answer in your notebook the following questions
- Another word for freezing is _____________.
- Other words for vaporisation are _________ and _________.
- The melting point is to a solid what the _________ point is to a liquid.
- For a solid to melt, it _________ heat.
- An example of subtance that sublimes is ____________.
- When a gas condense it __________ heat.
- The freezing point of a liquid is equal to the ________ point of a ________.
- Use an online Interactive Periodic Table (such as the one given above) to find the melting and boiling point of the element carbon.
3. the particle theory of matter
More than 2000 years ago in ancient Greece, a philosopher called Democritus suggested this hypothesis:
All matter, living and non-living, is made of tiny particles too small to be seen. His idea was that if you kept cutting something into smaller and smaller pieces, you would eventually come to the smallest possible particles—the building blocks of matter. He used the word atomos (which in Greek means ‘cannot be divided’) to describe these tiniest particles. This is where the word ‘atom’ comes from.
Over the years, scientists have carried out a lot of tests on matter and the results always agreed with Democritus hypothesis. When a hypothesis has been supported by a large amount of evidence and never contradicted, it becomes a theory. The particle theory of matter states that:
Using these simple statements, scientist were able to explain some of the most obvious physical properties of solids, such as their fixed shape, liquids, that adopt the shape of the container and gases that tends to occupy the maximum volume available. In the simplest interpretation, the differences between states of matter come down to the balance between attractive forces between particles and how fast they move depending on the temperature.
All matter, living and non-living, is made of tiny particles too small to be seen. His idea was that if you kept cutting something into smaller and smaller pieces, you would eventually come to the smallest possible particles—the building blocks of matter. He used the word atomos (which in Greek means ‘cannot be divided’) to describe these tiniest particles. This is where the word ‘atom’ comes from.
Over the years, scientists have carried out a lot of tests on matter and the results always agreed with Democritus hypothesis. When a hypothesis has been supported by a large amount of evidence and never contradicted, it becomes a theory. The particle theory of matter states that:
- All matter is made up of tiny particles too small to see.
- There are spaces between the particles.
- There are attractive forces between particles. The weaker these forces are, the further apart the particles are.
- The particles are always moving.
- At high temperatures the particles move faster than they do at low temperatures.
Using these simple statements, scientist were able to explain some of the most obvious physical properties of solids, such as their fixed shape, liquids, that adopt the shape of the container and gases that tends to occupy the maximum volume available. In the simplest interpretation, the differences between states of matter come down to the balance between attractive forces between particles and how fast they move depending on the temperature.
- In the solid state, particles stick together thanks to the strong attractive forces that exist between them. These forces that hold the particles together are called chemical bonds. Chemical bonds prevent particles from moving around. When particles are packed in a fixed, regular pattern, they form a crystal. Examples are ice, kitchen salt (sodium chloride) or a lump of copper. The diagram below is a model
- matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place.
- Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely.
- Matter in the gaseous state has both variable volume and shape, adapting both to fit its container. Its particles are neither close together nor fixed in place.
What is a model in science?
It is a simplified way of representing something that is too small to be seen (such as atoms or molecules) or too large or complicated to be studied easily (such as the solar system for example).
It is a simplified way of representing something that is too small to be seen (such as atoms or molecules) or too large or complicated to be studied easily (such as the solar system for example).
DID YOU KNOW?
The working principle of a fridge can be explained using the particle theory. A fridge works by removing heat from the inside of the fridge. To do that, a pump is used to force a liquid to vaporise when it flows in a pipe located inside the fridge. Since vaporisation requires heat to proceed, the liquid takes up heat from the surrounding air, which cools down the air. When the gas is recompressed outside the fridge, heat is being released, which is why the air behind a working fridge is always hot.
The working principle of a fridge can be explained using the particle theory. A fridge works by removing heat from the inside of the fridge. To do that, a pump is used to force a liquid to vaporise when it flows in a pipe located inside the fridge. Since vaporisation requires heat to proceed, the liquid takes up heat from the surrounding air, which cools down the air. When the gas is recompressed outside the fridge, heat is being released, which is why the air behind a working fridge is always hot.