elements and compounds
1. classification of matter
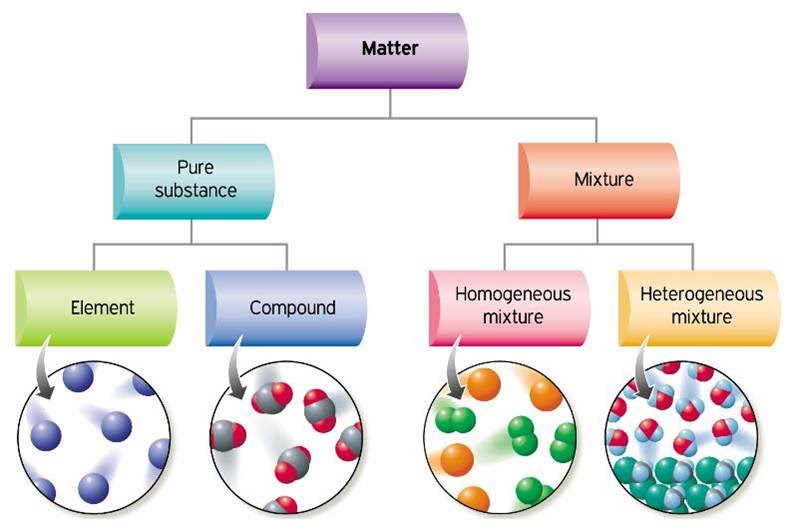
Any ordinary matter can be classified into the categories shown below.
A pure substance can be either an element or a compound.
Two or more pure substances that can be easily separated by physical means form a mixture. There can be two types of mixtures.
Click on the image above to obtain more information on the classification of matter.
- When all the atoms in a substance are alike, the substance is an ELEMENT. For example, the gas oxygen is made of oxygen atoms and a sample of mercury contains mercury atoms only.
- A COMPOUND is a substance with two or more elements combined in a fixed proportion. For example, chalk is a compound made of carbon, calcium and oxygen atoms and water is made of hydrogen and oxygen atoms. Kitchen salt consists of sodium and chloride ions, explaining why it is called sodium chloride.
Two or more pure substances that can be easily separated by physical means form a mixture. There can be two types of mixtures.
- A mixture of different and easily distinguishable materials, such as granite, concrete, and a noodle soup is a HETEROGENEOUS MIXTURE.
- A HOMOGENEOUS MIXTURE contains two or more gaseous, liquid, or solid substances blended evenly, for example air, salt water, or a steel (iron+carbon).
Click on the image above to obtain more information on the classification of matter.
2. Arranging the elements
|
Imagine searching through piles of unsorted CDs looking for the latest album of your favourite artist. Such a task could take days. Fortunately music shops arrange their CDs in separate sections. They then divide each of these groups into smaller groups according to the type of music, e.g. rock, popular, jazz and classical. Artists within each group are then arranged alphabetically.
Scientists have a similar problem to the music store owner. There are over 100 different elements, each with different physical and chemical properties. |
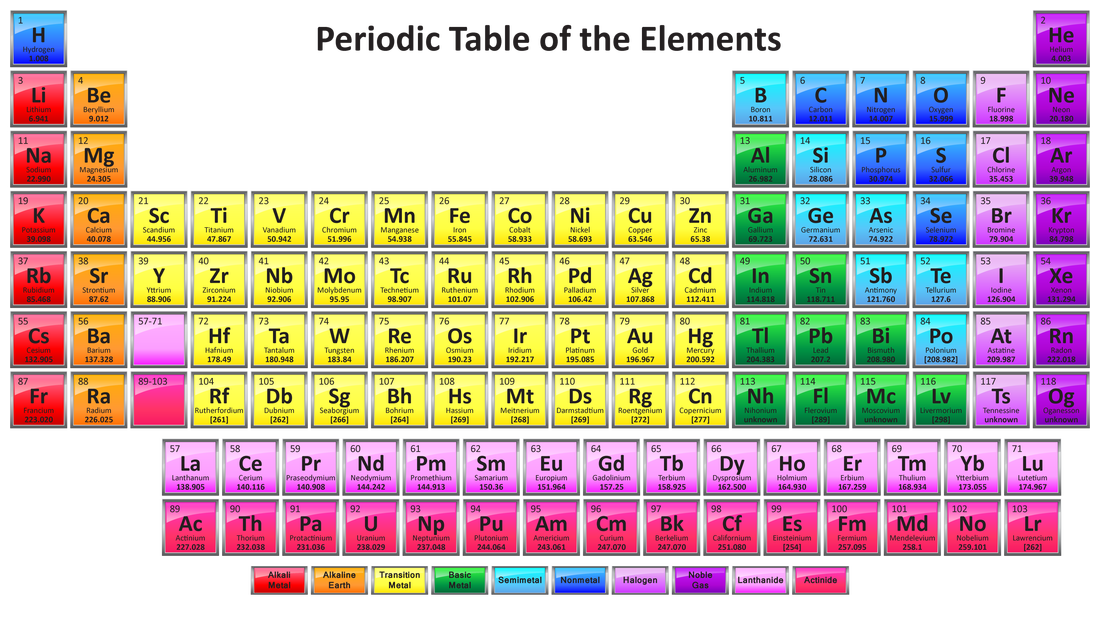
- The periodic table is simply a list of the chemical elements arranged in order of increasing atomic number. Reading like a book from left to right and top to bottom, the atomic number increases in single units from 1 to 118.
- The arrangement is such that similar elements are found next to one another in repeating cycles or periods. From this pattern comes the term 'The Periodic Table'. A number of trends in chemical and physical properties of the elements can be recognised and will be discussed in the next section.
- More than just a list of elements, the Periodic Table is one of the most useful datatables in all of science, providing information on the chemical and physical properties of elements, depending on their position in the table.
- The most common layout of the Periodic Table is shown below, although alternative layouts have been proposed. Click here to obtain more information on different presentations of the Periodic Table.
3. meaning of letters and numbers
|
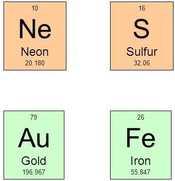
For convenience, each element has its own shorthand symbol. Look at the examples on the right handside.
The symbol of an element consists or 1 or 2 letter. The first letter is always written as a capital. The second letter is always written in small case. The symbol does not always tell what the element is, for example Au for gold or Fe for iron. |
The smallest of the two numbers (always a whole number) in the box is called the atomic number. It is equal to the number of protons. for example, the atomic number of neon is 10 because all neon atoms have 10 protons in their nucleus.
The largest number is called the average atomic mass, or atomic mass of the element.
The largest number is called the average atomic mass, or atomic mass of the element.
Activity: Try to write your first name or last name using symbols of the Periodic Table.
For example: Olivier can be made up of O, Li, V, I, and Er
For example: Olivier can be made up of O, Li, V, I, and Er
4. Structure of the Periodic Table
|
PERIOD: The horizontal rows of the Periodic Table are called periods.
There are 7 periods in the Periodic Table. The 2 rows placed at the bottom actually belong to the 6th and 7th periods. They have been removed from these rows to keep the Periodic Table compact. GROUP: The vertical columns of the Periodic Table are known as groups. There are numbered from 1 to 18. Some of the groups are also referred to by a name. The most well-known are: Group number 1: alkali metals Group number 17: halogens Group number 18: noble gases |
Elements of the same group share similar chemical properties owing to the similarity of electronic configuration in terms of the outer (valence) electron shell.
For example, elements of group 1 - the alkali metals - all have one electron in their outer shell. They are all soft metals, which react easily with water.
For example, elements of group 1 - the alkali metals - all have one electron in their outer shell. They are all soft metals, which react easily with water.