rate of reaction
In a nutshell
- Different reactions can happen at different rates. Reactions that occur slowly have a low rate of reaction.
- For a chemical reaction to occur, the reactant particles must collide. The rate of reaction depends on the proportion of effective collisions between reactants. The more effective collisions there are, the faster the rate of reaction.
- The rate of a chemical reaction depends on a number of factors: temperature of the reaction, concentration of the reactants, pressure of the reactants, surface area of one reactant, using a catalyst or stirring.
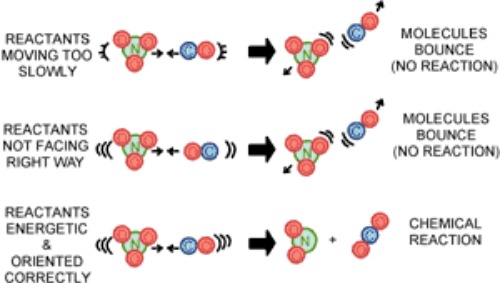
1. The collision theory
- For a reaction to occur, reactants must collide. The more often reactant molecules collide, the more often they react with one another, and the faster the reaction rate. If reactant particles do not collide with enough energy, they will not react together.
- In reality, only a small fraction of the overall collisions, are effective collisions, that result in a chemical reaction.
- This is because only a small number of molecules have the right orientation and with the right amount of energy at the moment of impact to break the existing bonds and form new ones.
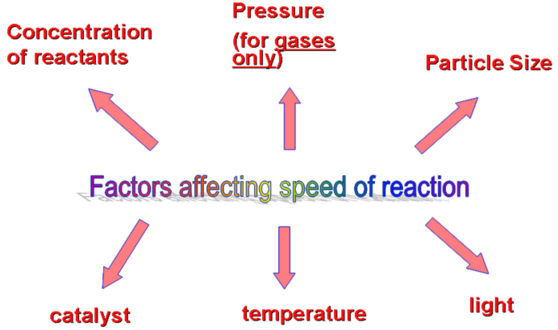
2. Factors affecting rates of reaction
The rate of a chemical reaction depends on a number of factors. The Collision Theory account for the way these factors work.
- Temperature: as the temperature increases, the rate of reaction increases.
- Concentration of the reactants: as their concentration increases, the rate of reaction increases.
- Pressure of any gaseous reactant: increasing the pressure of a gaseous reactant increases its concentration, therefore increase the rate of reaction.
- Particle size: Increasing the surface area of a solid reactant by increasing the size of particles increases the rate of reaction.
- Catalyst: Catalysts increase the rate of a reaction without being changed themselves by the end of the reaction.
- Light: The rate of some chemical reactions can be increased by using light with the correct frequency range, usually in the ultraviolet range. Such light break chemical bonds, therefore increasing the rate of reaction.
Here are more details on the way each factor acts on the speed of a chemical reaction
About temperature: temperature is a physical way to measure the amount of heat an object contains. At the atomic level, heat energy is due to the vibration and motion of atoms, molecules and ions making up any object. The faster the particles making up an object are moving and vibrating, the hotter the object feels. By moving faster, particles are more likely to collide and react. Have you already tried to bake cupcakes in an oven at 100 oC?
About concentration and pressure: Think about a class of year 8 students asked to run on the college's oval blindfolded for 5 minutes. Repeat the same experiment by changing the oval with an empty room the size of a classroom. In what situation will the students collide more often?
In that case, the number of students per square meter of ground is the concentration (in students per square meter) or the pressure. It is larger in the classroom where collision are more likely to occur than on the oval.
About surface area: Think about adding sugar to a cup of tea. You have the choice between white granulated sugar or sugar cubes. Which one will dissolve faster? Granulated sugar is expected to dissolve faster as it has more surface area available for the water to react. Or think about gun powder. It burns very well when it is finely divided as there is more room for the oxygen to react with the gunpowder grains. If it wasn't powdered, it would burn very slowly and would not produce the explosive effect wanted.
About a catalyst: If you want to meet your next girlfriend, you are more likely to meet her at a party than in the street. The party acts as a catalyst as it gather same minded people. That's why a catalyst is also called reaction helper.
About temperature: temperature is a physical way to measure the amount of heat an object contains. At the atomic level, heat energy is due to the vibration and motion of atoms, molecules and ions making up any object. The faster the particles making up an object are moving and vibrating, the hotter the object feels. By moving faster, particles are more likely to collide and react. Have you already tried to bake cupcakes in an oven at 100 oC?
About concentration and pressure: Think about a class of year 8 students asked to run on the college's oval blindfolded for 5 minutes. Repeat the same experiment by changing the oval with an empty room the size of a classroom. In what situation will the students collide more often?
In that case, the number of students per square meter of ground is the concentration (in students per square meter) or the pressure. It is larger in the classroom where collision are more likely to occur than on the oval.
About surface area: Think about adding sugar to a cup of tea. You have the choice between white granulated sugar or sugar cubes. Which one will dissolve faster? Granulated sugar is expected to dissolve faster as it has more surface area available for the water to react. Or think about gun powder. It burns very well when it is finely divided as there is more room for the oxygen to react with the gunpowder grains. If it wasn't powdered, it would burn very slowly and would not produce the explosive effect wanted.
About a catalyst: If you want to meet your next girlfriend, you are more likely to meet her at a party than in the street. The party acts as a catalyst as it gather same minded people. That's why a catalyst is also called reaction helper.
3. Collision Theory and Particle Theory
The Particle Theory and the Collision Theory are sometimes confused. While the Collision Theory explains how the speed of chemical reaction can be acted upon, the Particle Theory is useful to understand the physical properties of the various states of matter (solid, liquid, gas), as seen earlier in this course
Reminder: The Particle Theory states that all matter consists of many, very small particles which are constantly moving. The degree to which the particles move is determined by the amount of energy they have and their relationship to other particles. The particles might be atoms, molecules or ions. The use of the general term 'particle' means the precise nature of the particles does not have to be specified.
Reminder: The Particle Theory states that all matter consists of many, very small particles which are constantly moving. The degree to which the particles move is determined by the amount of energy they have and their relationship to other particles. The particles might be atoms, molecules or ions. The use of the general term 'particle' means the precise nature of the particles does not have to be specified.