Physical vs chemical change
1. Physical and chemical properties
|
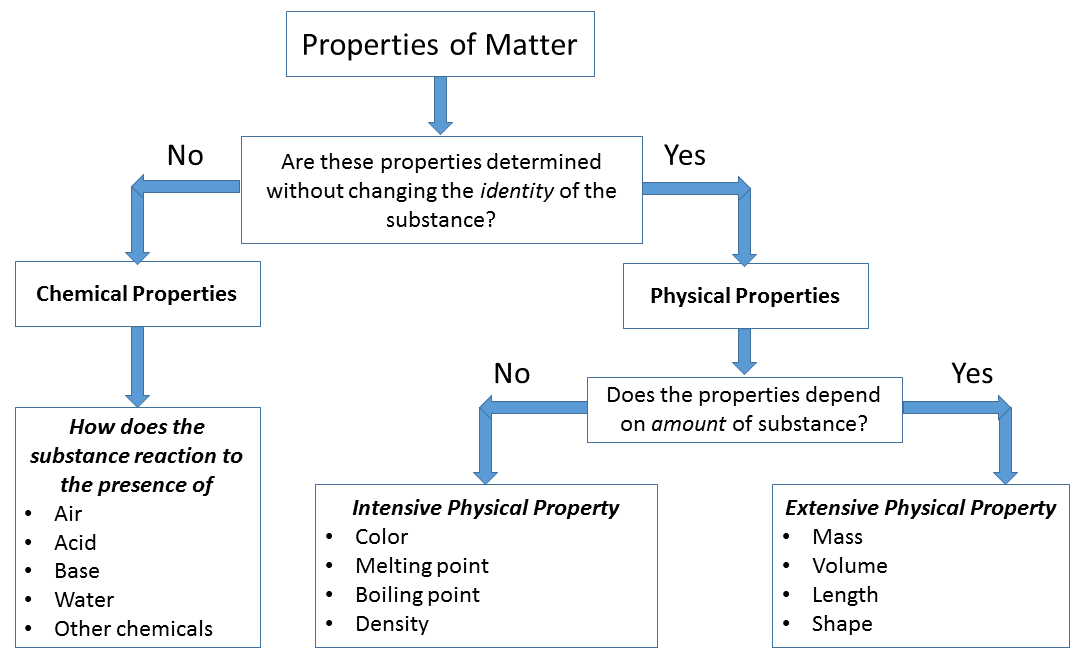
Physical properties
They are measured or observed without changing the identity of material. Examples: colour, melting point, boiling point, density, electrical conductivity |
Chemical properties
They are observed only after chemical change occurs. Examples: acidity or basicity, reactivity with water, combustibility, corrosion |
The flow chart below helps decide whether a material property is physical or chemical.
2. Physical or chemical change?
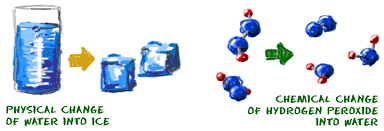
When a chemical change occurs, one or more new substances, called products, are formed. Examples are burning wood or rusting. In general, a chemical reaction cannot be easily reversed. You cannot get a log of wood back once you have burnt it!
When a physical change occurs, no new product is formed. A change of state may have occurred but the substance remains the same. A classical example is ice melting. Ice and liquid water are both made of the same molecules. An important characteristic of physical change is that it is generally reversible.
The section below summarises the differences between physical and chemical changes.
When a physical change occurs, no new product is formed. A change of state may have occurred but the substance remains the same. A classical example is ice melting. Ice and liquid water are both made of the same molecules. An important characteristic of physical change is that it is generally reversible.
The section below summarises the differences between physical and chemical changes.